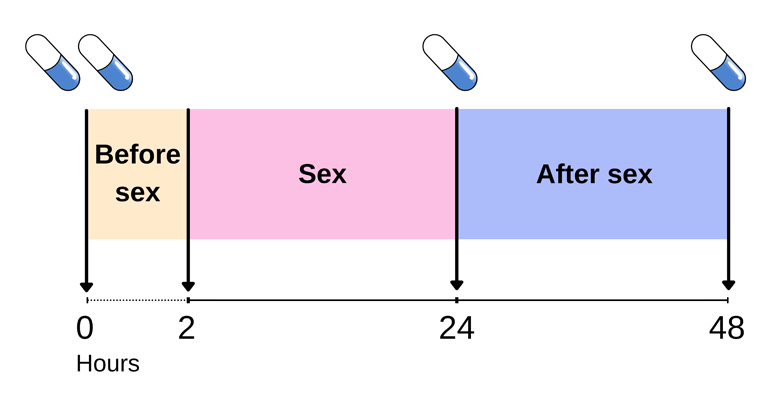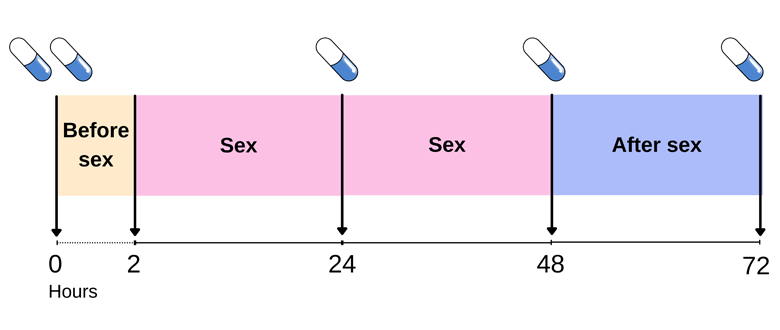
PrEP
"PrEP" stands for pre-exposure prophylaxis. It generally means the use of an antiretroviral medication to prevent the acquisition of HIV infection by uninfected persons. Two antiretroviral drugs for treatment of HIV infection were approved for use as PrEP, oral dose of co-formulated tenofovir disoproxil fumarate plus emtricitabine (TDF/FTC) and tenofovir alafenamide plus emtricitabine (TAF/FTC). There are also ongoing studies of other antiretrovirals for prevention, including injectable and long acting agents. As preventive agents administered prior to exposure, they are collectively called pre-exposure prophylaxis (PrEP).
As at 2021, oral tenofovir disoproxil fumarate (TDF) 300 mg / emtricitabine (FTC) 200 mg is the drug most often adopted in countries with PrEP programmes. Unless otherwise stated, PrEP in this FAQ will henceforth refer to the administration of oral TDF/FTC to HIV negative individuals for the sole purpose of preventing acquisition of HIV.